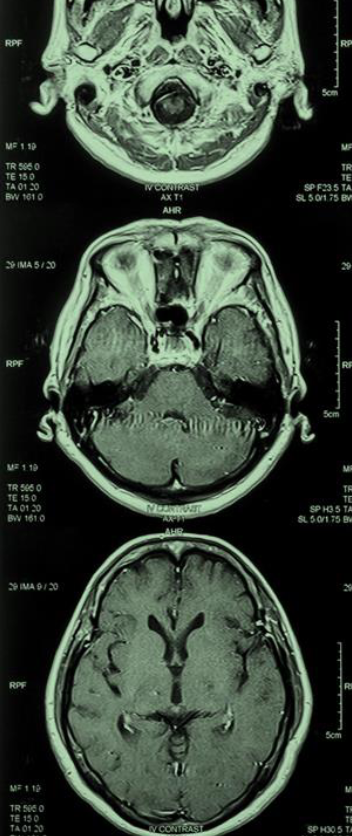
The field of Alzheimer’s disease (AD) research and treatment seems better primed than ever to deliver new therapies with the potential to transform care. The FDA’s approval of aducanumab in June 2021, though contentious, marks the first authorization of a therapy that targets the fundamental pathophysiology of the disease. More than 100 disease-modifying therapies are now in clinical trials–nearly 20 of which are in phase 3.3 The identification of biomarkers for AD has created new avenues for scientific exploration and increased hope that AD will one day be a treatable condition.
More than 6.5 million Americans are living with AD – a number projected to more than double by 2050 as the population ages. Without treatment, affected individuals face a progressive and ultimately lethal neurodegenerative disorder that destroys memory and thinking skills necessary to carry out simple daily tasks. As case numbers rise, healthcare and other societal costs associated with AD are predicted to reach $1.1 trillion.